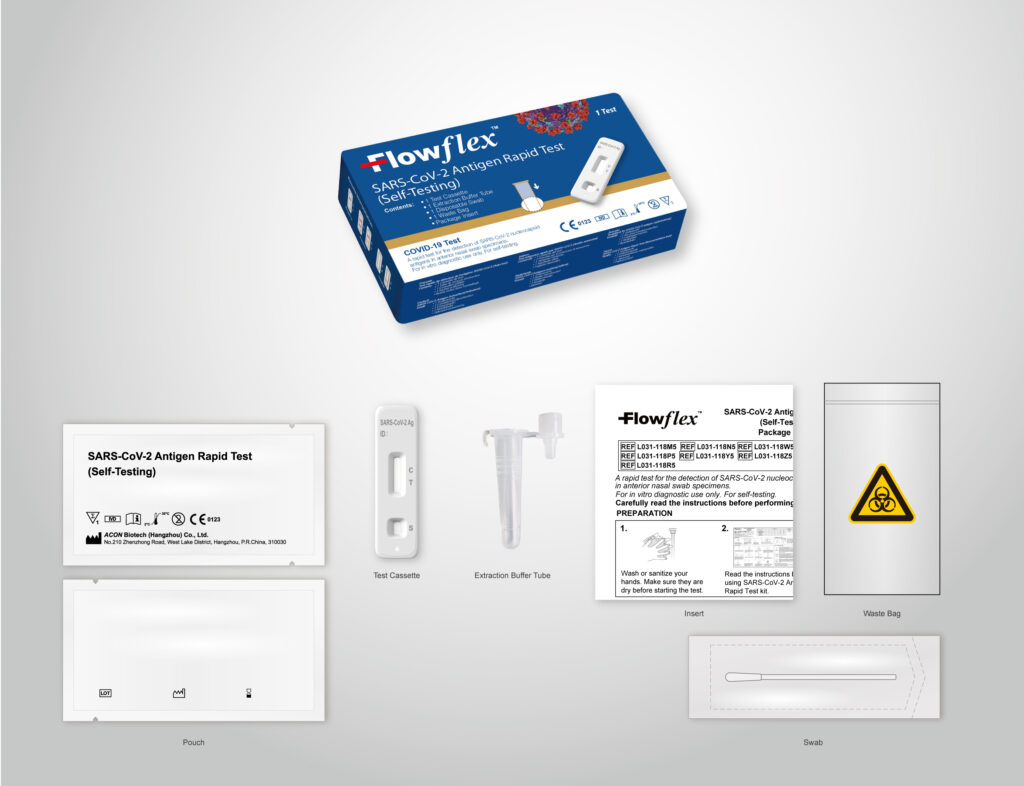
ACON Laboratories, Inc., a leading global medical device manufacturer for 25 years, announced today that its Flowflex™ COVID-19 Antigen Home Test has been authorised for emergency use (EUA) by the U.S. Food and Drug Administration.
The Flowflex COVID-19 Antigen Home Test is a simple nasal swab test which will soon be available for purchase without a prescription in major retail stores and online. It may be used for self-testing by individuals aged 14 years and older, or with adult-collected nasal swabs from children as young as 2 years old.
In contrast to other home tests which require testing twice within a two-to-three-day period (a process known as serial screening), the Flowflex COVID-19 Antigen Home Test has been authorised for use as a single test by individuals with or without symptoms. This will allow for the distribution of more affordable single-test packaging, resulting in greater access to home testing.
Flowflex COVID-19 tests are already available in many countries outside the U.S., including widespread distribution in the UK through the National Health Service (NHS). The international popularity of this test has led ACON to greatly expand global production capacity at multiple manufacturing sites. This emergency use authorisation will now allow ACON to quickly respond to the unmet demand for simple and inexpensive home diagnostics as a critical tool in the fight against COVID-19.
Download the FDA EUA approval letter HERE.
Source: Flowflex™ COVID-19 Home Test Receives FDA EUA – ACON LABS INC.