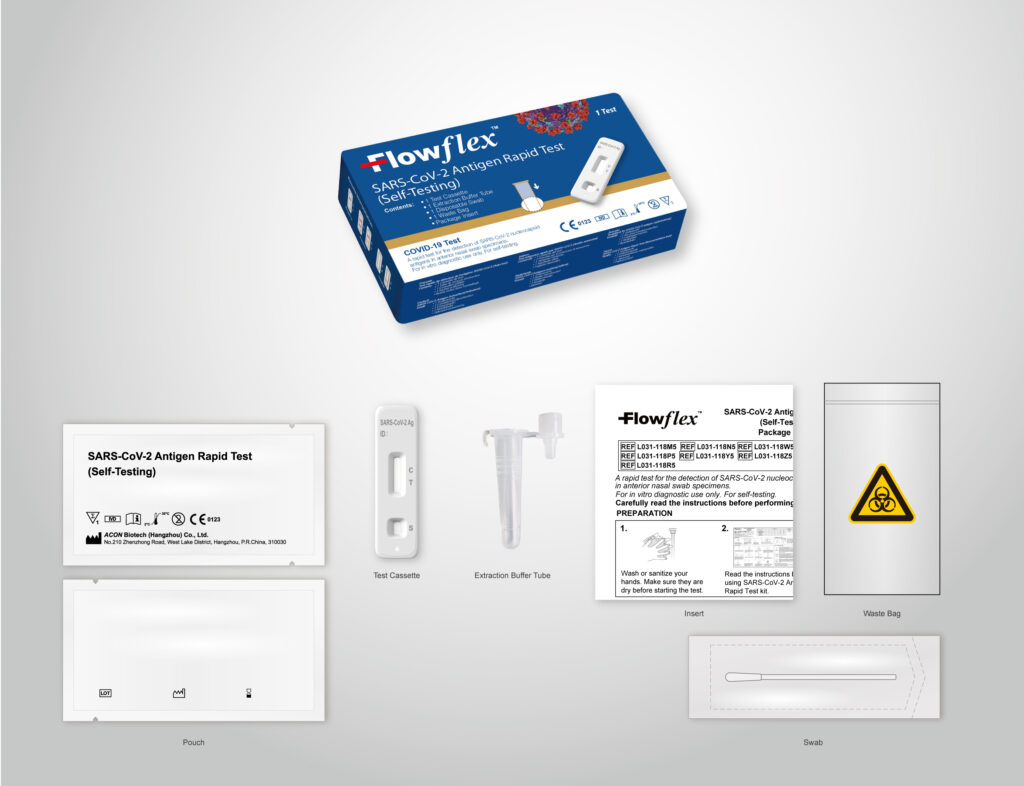
Flowflex SARS-CoV-2 Antigen Rapid Test has been authorised for as a self-test by the Australian Government Department of Heath Therapeutic Goods Administration (TGA).
The TGA’s performance requirements for COVID-19 rapid antigen self-tests are internationally aligned with technical specifications published by the World Health Organisation (WHO), and the European Commission. This includes a clinical sensitivity of at least 80% (for specimens collected within 7 days of symptom onset), and a clinical specificity of at least 98%.
The TGA has assessed the sensitivity of each test approved test, with Flowflex being categorised as “Very High Sentsitivity”, which equates to a clinical sensitivity greater than 95% PPA . This is based on the assessed studies carried out by the manufacturer which show the positive percent agreement (PPA). PPA is the proportion of individuals who tested positive to COVID-19 using a rapid antigen self-test, compared to those who tested positive to COVID-19 using a more sensitive PCR test.
The Flowflex SARS-CoV-2 Antigen Rapid Test is a simple nasal swab test that can used for self-testing.
Flowflex SARS-CoV-2 Antigen Rapid Tests are already available in many countries outside the Australia, including widespread distribution in the UK through the National Health Service (NHS).
To find out more visit the product page HERE, or for enquiries and orders please submit the contact form below, or call the team on +44 (0) 114 2213344.