Description
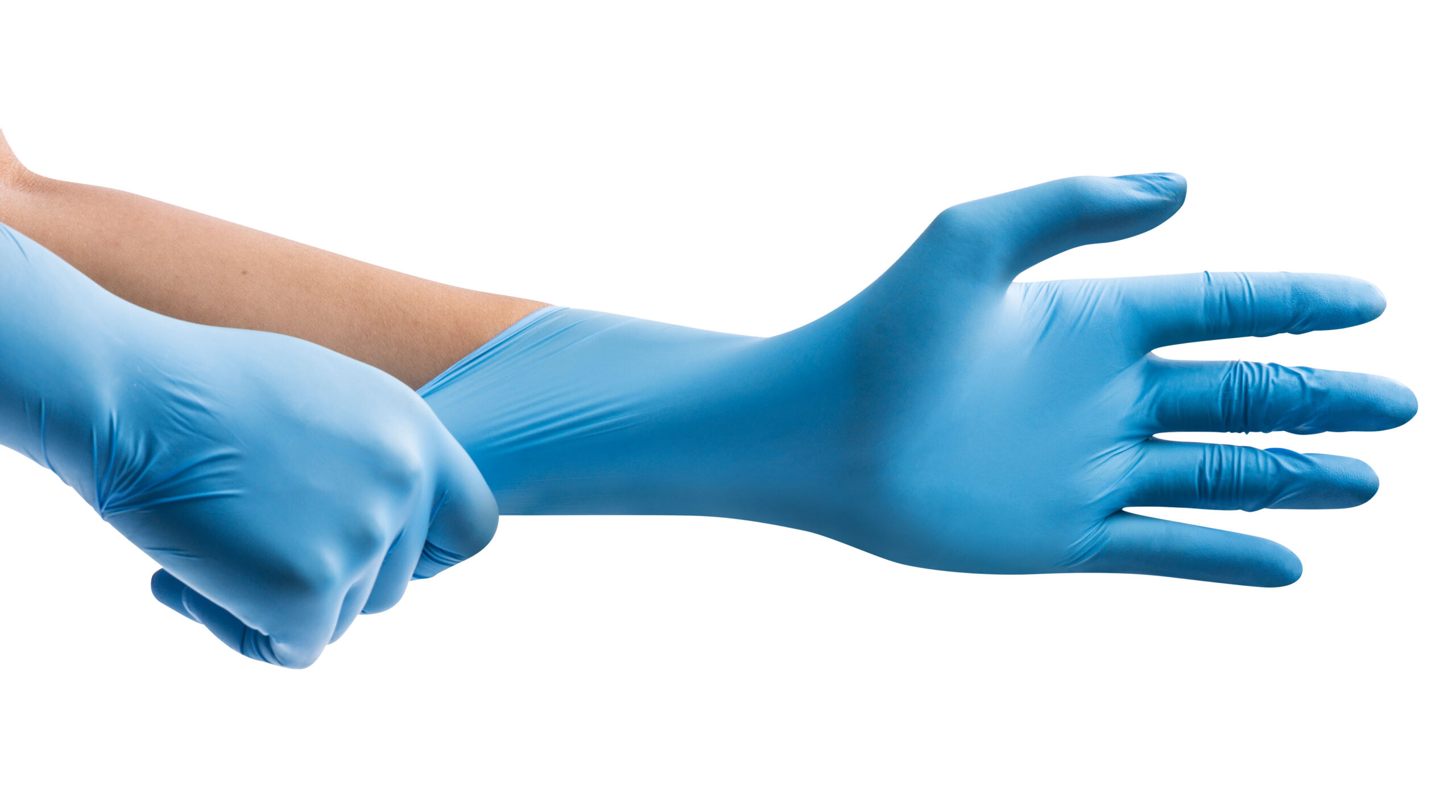
HAIKA® NX510 are lightweight, ambidextrous, single-use medical nitrile examination gloves.
They offer healthcare professionals high performance barrier protection as well as supreme comfort and touch sensitivity.
The glove is certified for medical use, whilst also having the necessary cerification to make it safe for use in and around food contact settings.
Secure Control
A textured surface enhances slip resistance and allows for a secure grip.
Supreme Comfort and Sensitivity
The lightweight and elastic nature of the nitrile deliversan excellent sense of touch and a comfortable wearing experience even when worn for long periods.
Effective Barrier Protection
Helps to protect users and patients from the transmission of infections.
Beaded Cuff
Durable beaded cuff design enables the glove to be donned and removed efficiently and securely.
Powder-free and Latex-free
Eliminates powder-induced irritation and makes it ideal for people with a Type I latex sensitivity.
Minimises the Risk of Allergic Contact Dermatitis
Free of Thiurams, Mercaptobenzotiazoles, Thiazoles and Thioureas.
Chemical Resistant
Tested in accordance with EN374-1:2016 for resistance to chemical substances.
Tested against chemotherapy drugs according to ASTM D6978-05 (2019).
The Haika NX510 is a 3.5 gram, 240mm long blue powder free nitrile glove. Boxed in 100’s the glove is silicone-free, powder-free, and latex-free – offering superior strength whilst maintaining maximum dexterity. The Haika NX510 has a microtexture with additional texture on the fingertips for improved pinch grip, a rolled cuff, is AQL 1.5, and touchscreen-friendly.
Specifications:
- 3.5 gram
- 240mm long
- Microtexture with additional texture on the fingertips for improved pinch grip
- Touchscreen compatible
- Durable beaded cuff
- Blue
- Powder-free
- Latex-free
- Silicone-free
- AQL 1.5
- CE certiified
- Class I certified
- CAT III certified
- AQL 1.5
- EN374-1 Type B
- EN374-5 Virus
- EN420
- EN455
- 5 years expiry from date of production
Sizes:
- S-Small
- Small
- Medium
- Large
- X-Large
- XX-Large
Packaging:
- 100 gloves per box/dispenser
- 10 boxes/dispensers per case